Same Day Shipping. Quick Response. Call us at 800.232.2403.
NexScreen Rapid Urine Drug Test Cups
The NexScreen Rapid Urine Drug Test Cup offers the best value of any drug screening cup. We have multiple drug configurations that include testing for the newest trends in drug abuse. They are accurate, easy, rugged, and flexible.

Shop Products
Tests
# of Panels
-
NexScreen 6 Panel Drug Screen Cup
CLIA-Waived: AMP500, COC150, MET500, OPI2000, PCP25, THC50SKU: ND-60610N$137.50 -
NexScreen 10 Panel Drug Screen Cup
CLIA-Waived: AMP500, BAR300, BZO300, COC150, MDMA500, MET500, MTD300, OPI2000, OXY100, THC50SKU: ND-61012N$153.75 -
NexScreen 12 Panel Drug Screen Cup
AMP1000, BAR300, BZO300, COC300, MDMA500, MET1000, MTD300, MOP300, OXY100, PCP25, TCA1000, THC50SKU: 61203N$175.00 -
NexScreen 13 Panel Drug Screen Cup
CLIA-Waived: AMP500, BAR300, BZO300, COC150, MDMA500, MET500, MTD300, OPI300, OXY100, PCP25, TCA1000, THC50, BUP10SKU: ND-60135N$181.25 -
UScreen 12 Panel Drug Screen Cup w/Adulterants
THC50, COC300, MOP(OPI300), AMP1000, MET1000, BAR300, BZO300, PCP25, MDMA500, MTD300, OXY100, TCA1000 (CR,SG,PH)SKU: USSCUPA-12CLIA$181.25 -
5 Panel Drug Screen Cup w/Adulterants
BZO300, COC300, MET1000, OPI2000, THC50, Adulterants (OX/SG/pH)SKU: HCDOAV-654A3$137.50 -
5 Panel Clicker Drug Screen Cup (No THC)
AMP1000, COC300, MET1000, MOP300, OXY100SKU: HCDOAV-855$127.50 -
7 Panel Clicker Drug Screen Cup (No THC)
AMP1000, BAR300, BZO300, COC300, MOP300, MTD300, OXY100SKU: HCDOAV-975$150.00 -
12 Panel Clicker Drug Screen Cup
AMP1000, BAR300, BUP10, BZO300, COC300, MDMA500, MET1000, MOP300, MTD300, OXY100, PCP25, THC50SKU: HCDOAV-6125$175.00 -
13 Panel Clicker Drug Screen Cup
AMP1000, BUP10, BZO300, COC300, ETG500, FEN10, MDMA500, MET1000, MOP300, MTD300, OXY100, THC50, TRA200, Adulterants (CR/SG/pH)SKU: HCDOAV-8135EF1TA3A$181.25 -
14 Panel Clicker Drug Screen Cup
AMP1000, BAR300, BUP10, BZO300, COC300, EDDP300, MDMA500, MET1000, MOP300, MTD300, OXY100, PCP25, TCA1000, THC50SKU: HCDOAV-2145$187.50 -
14 Panel Clicker Drug Screen Cup
AMP1000, BAR300, BUP10, BZO300, COC300, ETG500, FEN10, MDMA500, MET1000, MOP300, MTD300, OXY100, PCP25, THC50SKU: HCDOAV-6145EF1$187.50 -
14 Panel Clicker Drug Screen Cup
AMP1000, BUP10, BZO300, COC300, ETG500, FEN20, K2(50), MDMA500, MET1000, MOP300, MTD300, OXY100, THC50, TRA200SKU: HCDOAV-8145EF1KT$187.50 -
16 Panel Clicker Drug Screen Cup
AMP1000, BAR300, BUP10, BZO300, COC300, ETG500, FEN300, K2(50), MDMA500, MET1000, MTD300, OPI2000, OXY100, PCP25, THC50, TRA200SKU: HCDOAV-6164EF1KT$200.00 -
DOT Urine Drug Screen Full Service
DOT Urine Drug TestSKU: DOT-8181$69.00

- FDA 510K cleared, CLIA-waived
- As accurate as a lab test
- Results in 5 minutes or less
- Multiple configurations in stock and ready to ship
- No Paper CCF when used with NexScreen Integrated Solutions
Accurate Results in Just 5 Minutes

Collect
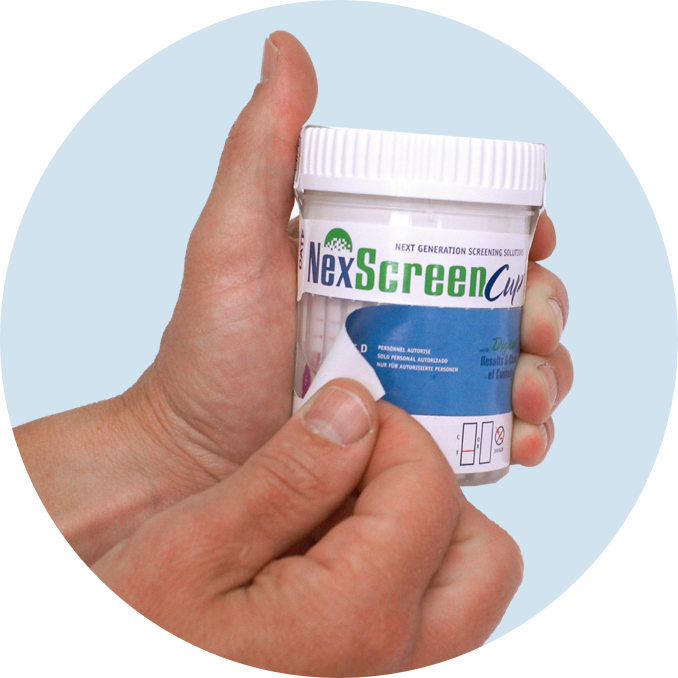
Peel

Read
NexScreen Integrated Solutions

NexScreen Integrated Solutions optimizes your POCT program. The advanced electronic screening management system provides a web-based portal for managing all stages of a drug test transaction. Reduce manual work and increase efficiency using electronic collection, automatic reporting, and fully digital chain of custody and control forms.
- No More Paper CCFs
- Digital Signatures for Donor & Collector
- See All Stages in One View
- Real Time Status Updates
- Expedited MRO Adjudication Process
Downloads
Fact sheets and other helpful documents.
FAQ's
Can’t find the information you need? We’d be happy to help. Just send us a message or call 888.956.8989.
No, store the product at room temperature up to the date of expiration printed on the individual foil pouch. Do not freeze. Do not store them in extreme heat or cold and keep them sealed in the foil pouch until use.
Collect at least 30ml of specimen.
The test results are valid up to 8 minutes after the test has been administered.
Yes, once a cup has been voided in by the donor devices should be disposed of in a biohazard container. Please check with your state ordinance regarding disposal procedures. Urine specimens may be infectious; properly handle and dispose of all urine and urine reaction devices in a biohazard container.
There are a handful of products, called adulterants, which some donors use to attempt to change the result of a test. However, we offer up to six validity screens that detect adulterants.
The NexScreen Cup provides only the initial preliminary screening result. It will be necessary to confirm a presumptive positive result in a lab to conclude a test is positive. NexScreen strongly recommends that a SAMHSA‑certified laboratory confirms all non-negative results. Please call your distributor for more information.
All of our products have FDA 510k clearance.
No formal training is required. To provide your collectors with confidence on their first collection, NexScreen provides a complimentary certification exam under our Resources page.

